Abstract
Severe congenital neutropenia (SCN) is characterized by persistent neutropenia and risk of invasive, life-threatening infection as well as transformation to hematopoietic malignancy. The closely related syndrome cyclic neutropenia is characterized by recurrent episodic neutropenia accompanied by symptoms including infection. Understanding the genetic etiology of congenital neutropenia can help to direct therapy, guide surveillance and health maintenance strategies, and contribute to our understanding of basic neutrophil biology. Although mutations in ELANE are the most frequent cause of congenital neutropenia, there is a wide and ever-growing list of additional causative variants. Additionally, there appears to be regional genetic variability. For example, mutations in HAX1 are rarely if ever observed in North America while they are more common in Europe. We undertook exome sequencing of a large cohort from the Severe Chronic Neutropenia International Registry (SCNIR) of North America in an effort to define the genetic spectrum of congenital neutropenia and aid in the discovery of new pathogenic variants.
We expanded our previously reported study of whole exome sequencing to include 152 cases of chronic neutropenia, comprised of 94 cases of SCN and 58 cases of cyclic neutropenia. We selected cases in which ELANE testing was negative, or in a small minority of cases, where ELANE testing had not yet been performed. Indeed, exome sequencing only identified 7 cases (5 SCN and 2 cyclic) carrying pathogenic ELANE mutations in this cohort. In the remaining 145 cases, we analyzed exomes for the presence of variants in genes previously associated with congenital neutropenia including AK2, AP3B1, CD40LG, CLPB, CSF3R, CXCR2, CXCR4, DNAJC21, DNM2, DOCK2, EFL1, EIF2AK3, ELANE, G6PC3, GATA1, GATA2, GFI1, GINS1, HAX1, IRAK4, JAGN1, KAT6A, KRAS, LAMTOR2, LYST, MYD88, PGM3, PSTPIP1, RAB27A, RAC2, SBDS, SEC61A1, SLC37A4, SMARCD2, SRP54, STK4, TAZ, TCIRG1, TCN2, TLR8, USB1, VPS13B, VPS45, WAS, WDR1 and WIPF1. Pathogenic heterozygous mutations of CLPB that localize to the ATP-binding pocket were identified in 7 cases, making it the second most common cause of congenital neutropenia in North America. We additionally identified 4 cases with G6PC3 pathogenic variants, and one case each with pathogenic variants in JAGN1, CXCR4 (the cause of WHIM syndrome), germline homozygous CSF3R, and GFI1.
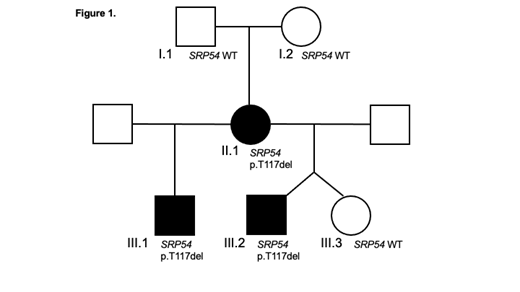
Interestingly, we identify 2 unrelated individuals (one with SCN and one with cyclic) and 2 siblings with SCN all of whom possess a recently described heterozygous variant in SRP54 (p.T117del). We collected genomic DNA from the affected mother of the 2 siblings, an additional unaffected sibling, and the unaffected grandparents. Through this kinship, we can confirm the de novo appearance of this variant in the second generation and demonstrate that it tracks with disease status (Figure 1). We also identified 2 unrelated individuals with SRP54 variants affecting residue 175 also located within the GTPase domain (p.G175E or p.G175del). Both variants are absent from the gnomAD database, and studies are underway to demonstrate de novo acquisition.
In summary, we have defined the spectrum of mutations present in ELANE-wildtype chronic neutropenia cases in North America. Pathogenic or likely pathogenic variants were identified in 26 out of 145 (18%) cases. The most frequently mutated genes were of CLPB, SRP54, and G6PC3, while mutations in HAX1 were not seen. Importantly, some of these mutations are associated with genetic syndromes with extra-hematopoietic findings (for example, CLPB and SRP54) that would warrant additional evaluations and targeted health maintenance. These findings emphasize the importance of sending large panels for genotyping, rather than targeted ELANE testing.
Bolyard: X4 Pharmaceuticals: Research Funding. Makaryan: Emendo Biotherapeutic: Research Funding. Dale: X4 Pharmaceuticals: Consultancy, Honoraria, Research Funding.
Author notes
 This icon denotes a clinically relevant abstract
This icon denotes a clinically relevant abstract

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal